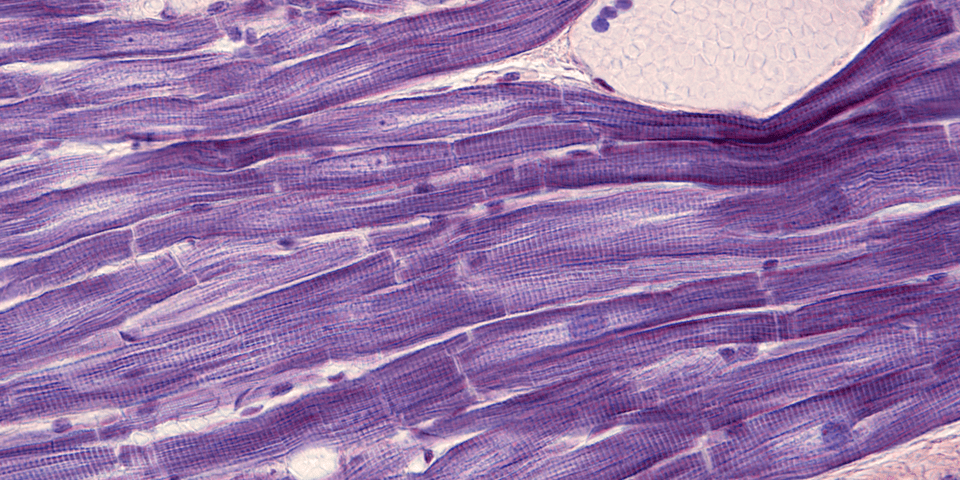
Myocardial fibrosis is a progressive accumulation of extracellular matrix (ECM) proteins that are produced by activated fibroblasts or by fibroblast-derived myofibroblasts. This reduces tissue compliance and accelerates the progression to heart failure (HF). Currently, therapies targeting angiotensin II and mineralocorticoid receptors are used in the clinics and reduce development of myocardial fibrosis. However, more efficient therapies are needed to prevent or even reverse myocardial fibrosis and progression of HF. A group of scientists at the Research Unit of Biomedicine, Department of Pharmacology and Toxicology, University of Oulu (Finland), aimed to identify novel miRNAs that regulate development of myocardial fibrosis and could serve as potential targets of therapy for myocardial fibrosis.
Muscular Dystrophy Research – Challenges and Breakthroughs
April 4, 2022
World’s First Spinal Cord Injury Treatment Using iPS cells
March 7, 2022Myocardial Fibrosis
Fibrosis is an established morphological feature of the structural myocardial remodeling that occurs in several cardiac diseases. This feature confers an increased risk for adverse cardiovascular events such as ventricular dysfunction and arrhythmias. Myocardial fibrosis targets and biomarkers under investigation include proteins, proteoglycans, and microRNAs (miRNAs). Characterization of individual miRNAs or miRNA expression profiles that are specifically associated with myocardial fibrosis might allow researchers to develop diagnostic tools and innovative therapies for fibrogenic cardiac diseases.
MicroRNA
MicroRNAs (miRNAs) are a class of small non-coding RNAs, which function post-transcriptionally by complimentary binding to their target mRNAs. Some miRNAs have been shown to regulate cardiac fibroblast function and myocardial fibrosis in experimental models, whereas their role in the human heart is poorly understood. Since a single miRNA can control the expression of multiple genes, they offer an attractive target for therapy as modulation of a single miRNA could have a broader effect on genes underlying the given pathology. MiRNAs can regulate the development of cardiac fibrosis through diverse aspects, e.g. miR-21 affecting fibroblast survival, miR-29 family controlling ECM protein expression, miR-125b governing fibroblast activation and miR-30 targeting connective tissue growth factor (CTGF) to control fibrotic remodeling.
The team at the Research Unit of Biomedicine, has found that miR-185-5p promotes collagen deposition and myocardial fibrosis in experimental models and identify an increase in miR-185-5p levels in cardiac samples of patients with end-stage cardiomyopathy. MiR-185-5p regulates collagen production by targeting apelin receptor. This novel mechanism may contribute to the development of myocardial fibrosis and pathological LV remodeling.
They used Collagen Staining Kit (Sirius Red/Fast Green) from AMSBIO, for detection of collagen deposition. Sirius Red/Fast Green Collagen Staining showed that mimic-185-5p augmented total collagen deposition in hCFs compared to control mimic treated cells. On the other hand, downregulating miR– 185-5p with antimiR-185-5p significantly attenuated collagen deposition. Studies in fibroblast model show that miR-185-5p promotes collagen deposition, whereas targeting miR-185-5p attenuates collagen production.
Does the microRNA (miR-185-5p) regulate myocardial fibrosis?
The team at the Research Unit of Biomedicine, Department of Pharmacology and Toxicology, University of Oulu, highlighted the following:
- Analysis for cardiac samples from sudden cardiac death victims with extensive myocardial fibrosis as the primary cause of death identified dysregulation of miR-185-5p.
- Analysis of resident cardiac cells from mice subjected to experimental cardiac fibrosis model showed induction of miR-185-5p expression specifically in cardiac fibroblasts.
- In vitro, augmenting miR-185-5p induced collagen production and profibrotic activation in cardiac fibroblasts, whereas inhibition of miR-185-5p attenuated collagen production.
- In vivo, targeting miR-185-5p in mice abolished pressure overload induced cardiac interstitial fibrosis.
- Mechanistically, miR-185-5p targets apelin receptor and inhibits the anti-fibrotic effects of apelin.
- Finally, analysis of left ventricular tissue from patients with severe cardiomyopathy showed an increase in miR-185-5p expression together with pro-fibrotic TGF-β1 and collagen I.
- Overall, their work shows that miR-185-5p targets apelin receptor and promotes myocardial fibrosis
Further reading (published paper)
Lin, R., Rahtu-Korpela, L., Szabo, Z., Kemppi, A., Skarp, S., Kiviniemi, A. M., … & Kerkelä, R. (2022). MiR-185-5p regulates the development of myocardial fibrosis. Journal of molecular and cellular cardiology, 165, 130-140. Available via ScienceDirect: https://www.sciencedirect.com/science/article/pii/S0022282821002467