Muscular dystrophies are a group of inherited genetic diseases that cause muscle weakness over time. They result in disability or even death, and there is no cure for them. Many mutations that cause muscular dystrophies have been identified thanks to advances in genome sequencing. This has opened the door to new medicines that could regenerate the damaged muscles or repair incorrect genes. Even though, putting these theories to the test will necessitate more advanced methods for simulating real muscular dystrophy in the lab. Therefore, a group of scientists at the Department of Neurology, University of Massachusetts Chan Medical School, (USA) aimed to investigate therapeutic development for muscular dystrophies.

How to be GMP Ready: A Guide
August 31, 2022
MiRNA as a Novel Therapy for Myocardial Fibrosis
March 21, 2022Research Challenges
- Coaxing skin or other cells from an individual to become ‘induced pluripotent stem cells,’ which can then mature to become practically any adult cell in the body, including muscles, is one technique for modelling muscular dystrophy.
- However, this method rarely results in the formation of myoblasts, the ‘precursor’ cells that grow into muscle during development. This restricts research into how disease-causing mutations affect muscle development in the early stages.
Research Breakthrough
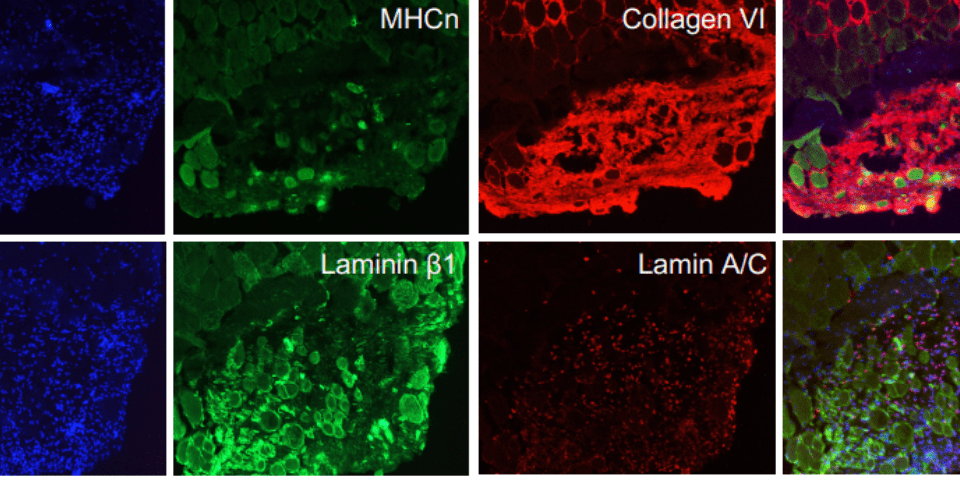
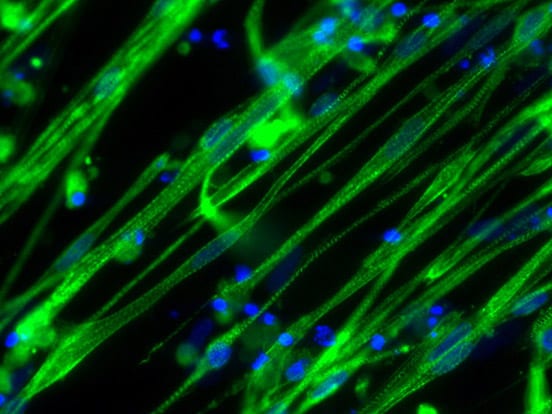
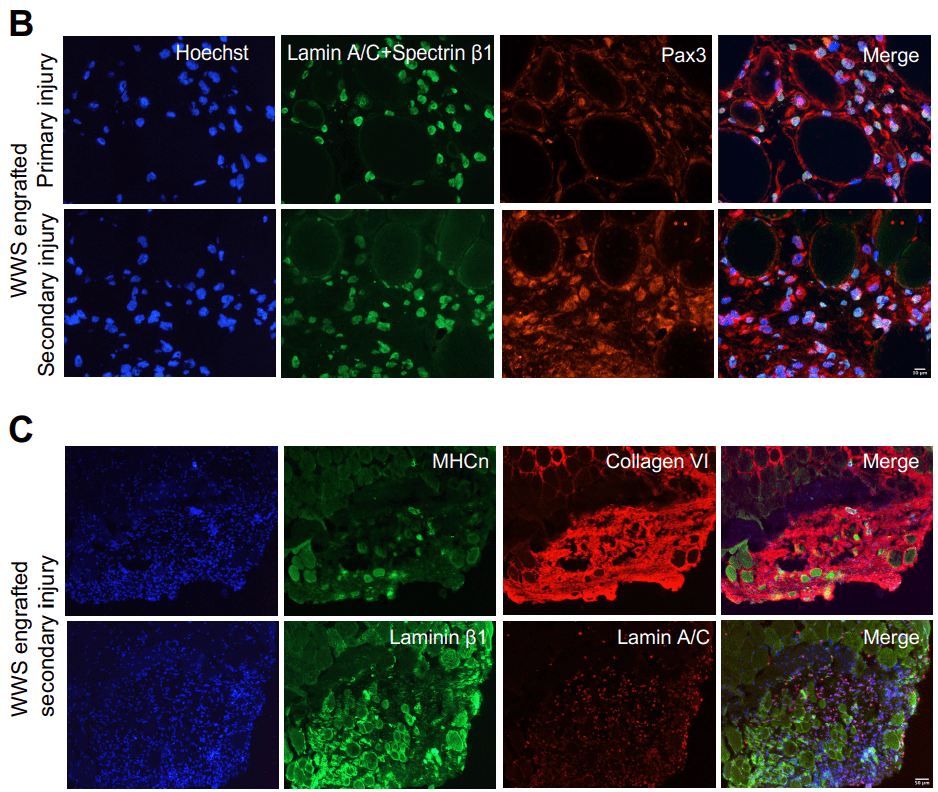
As a result, Charles Emerson and his team at the UMass Chan Medical School developed a two-step protocol for isolating and growing ‘induced myoblasts’ from induced pluripotent stem cells collected from healthy volunteers and muscular dystrophy patients (Guo et al. 2022). The team were able to model three different kinds of muscular dystrophy using these induced myoblasts, which can make more of themselves and become muscle. When these myoblasts were implanted into adult mouse muscles, they behaved like stem cells: some produced human muscle tissue, while others remained as precursor cells, ready to respond to muscle injury and begin healing.
All their culture and skeletal muscle myoblast (iMyoblast) isolation studies were performed using the skeletal muscle differentiation Kit from AMSBIO to differentiate human pluripotent stem cells into skeletal muscle. Explore skeletal muscle differentiation kit from AMSBIO here: https://www.amsbio.com/skeletal-muscle-differentiation/
To Sum Up
The findings by Guo et al. established the utility of skeletal muscle myoblasts (iMyoblasts) for ex vivo and in vivo investigations of human myogenesis and disease pathogenesis and for the development of muscle stem cell therapeutics. Thanks to their research discovery of the induced myoblasts, scientists will be able to explore and investigate the effects of diverse mutations on muscle tissue and better evaluate treatments. They could potentially be employed to repair muscle cells in patients as part of regenerative medicine treatments.
Further reading
- For further information on skeletal muscle differentiation kit from please visit: https://www.amsbio.com/skeletal-muscle-differentiation/ or contact AMSBIO on+44-1235-828200 / +1-617-945-5033 / [email protected].
- Guo, D., Daman, K., Chen, J. J., Shi, M. J., Yan, J., Matijasevic, Z., … & Emerson Jr, C. P. (2022). iMyoblasts for ex vivo and in vivo investigations of human myogenesis and disease modeling. eLife, 11, e70341. Available online at elife sciences: https://elifesciences.org/articles/70341
- Ps: Image in fig 1 is reproduced from Guo, D., et al. (2022). eLife, 11, e70341. iMyoblasts for ex vivo and in vivo investigations of human myogenesis and disease modeling. under a Creative Commons license (CC BY 4.0).