Benefits
- A superior low-attachment solution
- Easy to handle
- Excellent reproducible results
- Compatible with variety of cell-based assays
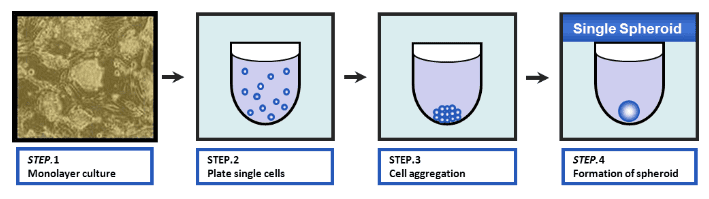
Spheroid and embryoid body (EB) cell culture is typically based on the spontaneous formation of an aggregate of cells in an environment where cell-cell interactions dominate over cell-substrate interaction. This can be achieved by using low-attachment cell culture conditions.
Lipidure® powder is a biocompatible and hydrophilic white powder which consists of 2-(methacryloyoxy)ethyl 2-(trimethylammonio)ethyl phosphate-n-butylmethacrylate copolymer. The building unit of this copolymer is 2-(methacryloyoxy)ethyl phosphorylcholine (MPC) monomer. Lipidure® mimics cell membrane surface and its molecular structure is the key for high hydrophilic nature and extremely low toxicity.
How Does Lipidure®-COAT Work?
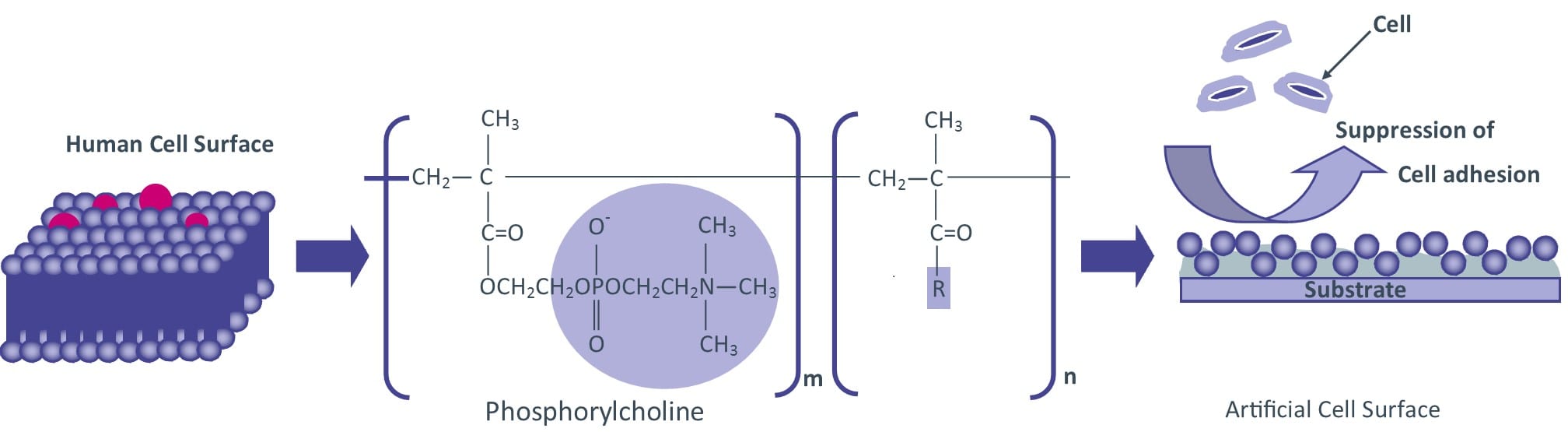
- Low-adhesion surface promotes cell aggregation & spheroid formation.
- Uses a biocompatible MPC Polymer containing Phosphoryl Choline (which is found in cell membranes).
- Completely synthetic, containing no substances of biological origin.
Formation of Single, Centrally Located Spheroid/EB on U-Bottom Plate
Cell Types Tested
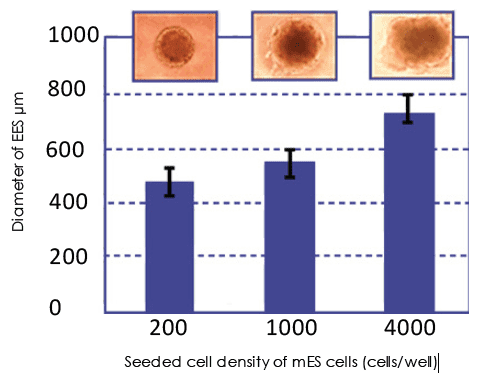
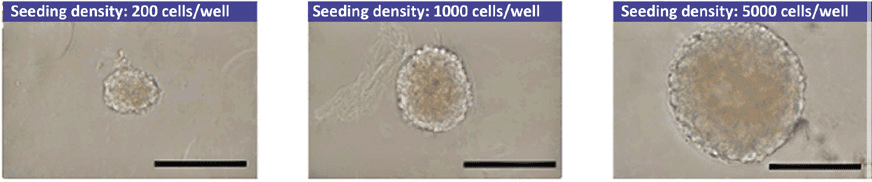
Spheroid formation using Lipidure® has already been demonstrated for the following cell types:
- human and mouse ES cells
- human and mouse iPS cells
- NIH3T3
- pre-adipocytes
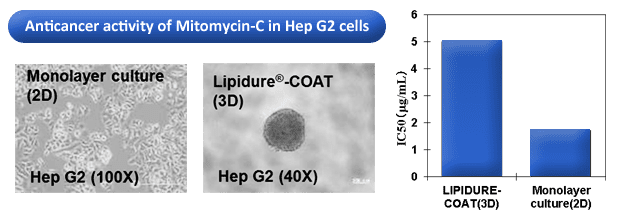
- HepG2 and other cancer cell lines
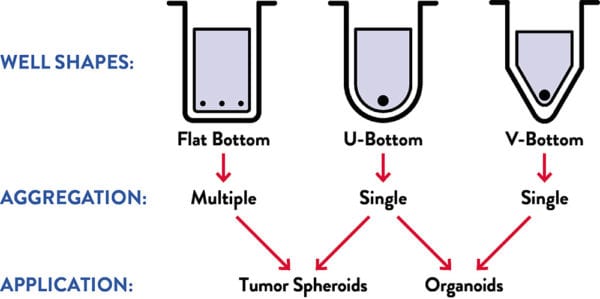
Applications
Choose your well shape for Lipidure®-Coat
Overall morphology of organoids in a culture can be altered significantly by whether cells are reaggregated in a V-bottom or U-bottom well (see Mellough et al 2019).
Superior Spheroid Formation
A single spheroid is generated in the Lipidure®-COAT U-bottom well, while numerous satellite spheroids are found in the competitor’s plates. This phenomenon shows that a lot of cells adhered to the competitor’s “low-adhesion” surface.

| Seeding Number (cells/well) | Diameter of Spheroid (μm) | |
|---|---|---|
| Lipidure-COAT | Competitor | |
| 1,000 | 185 ± 12 | 161 ± 9 |
| 10,000 | 423 ± 7 | 323 ± 46 |
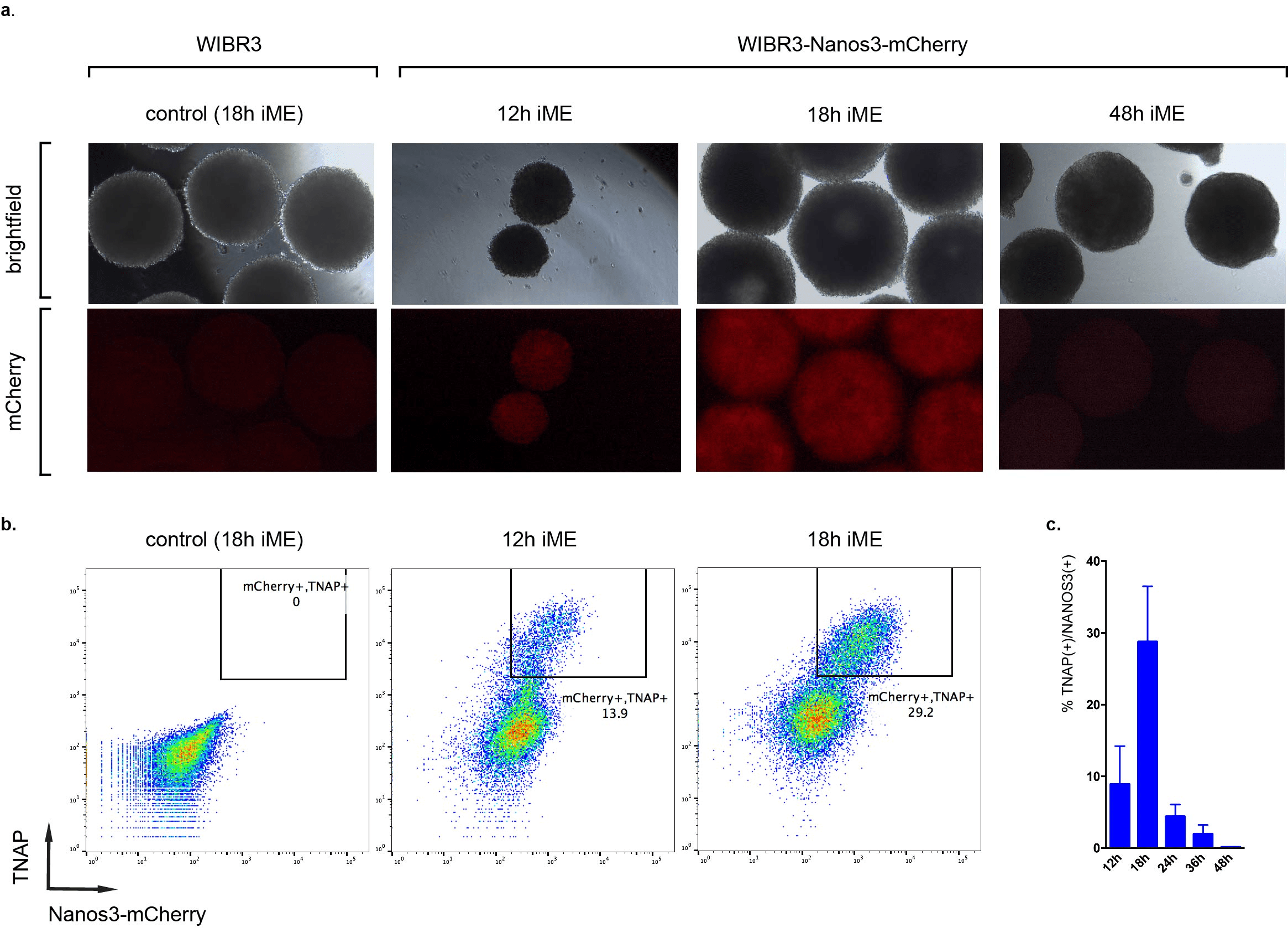
"We noticed a significantly reduced number of satellite spheroids and increased efficiency of differentiation with Lipidure U-bottom plates compared to other brands for the induction of primordial germ cell-like cells (PGCLCs)"
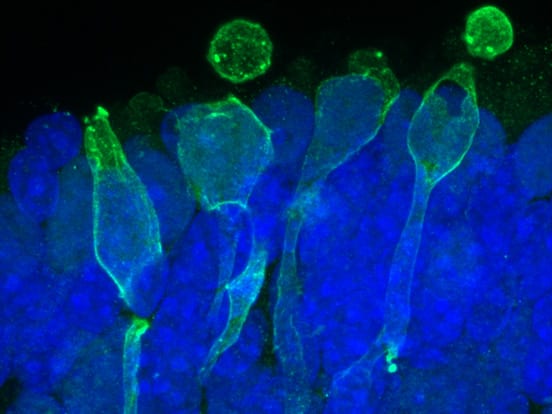
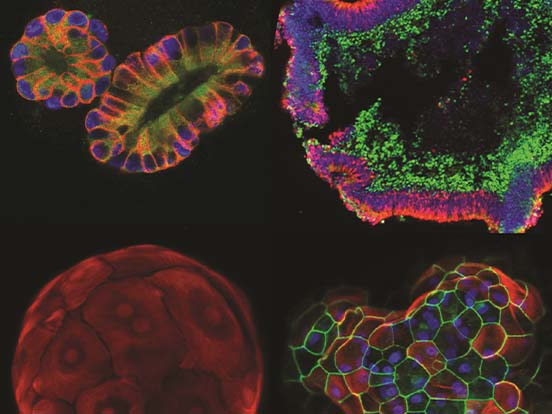
Organoid Culture
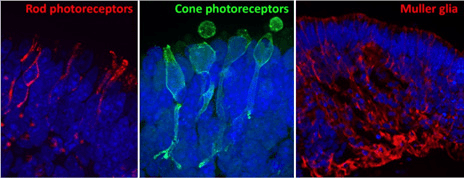
Images courtesy of Dr. Valeria Chichagova (Newcells Biotech) and Prof. Lako (Newcastle University).
Retinal organoids continuously grown on Lipidure-coated plates from the start of differentiation. Immunofluorescence images of 150 day old organoids show rod and cone photoreceptors containing outer segments on the apical side and synaptic terminals at the base. Muller glial cells span across the entire thickness of retinal neuroepithelium.
“The Lipidure-coated plates provided by AMSBIO were extremely useful for generating with ease large numbers of homogeneous retinal organoids which responded to light and contained all the key retinal cell types.”
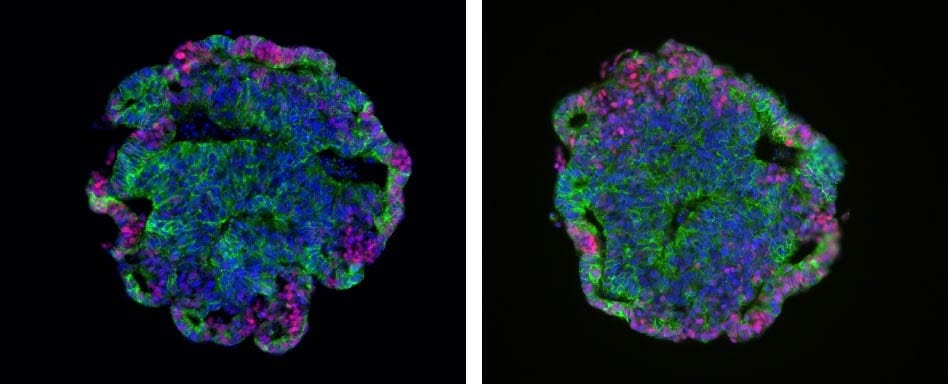
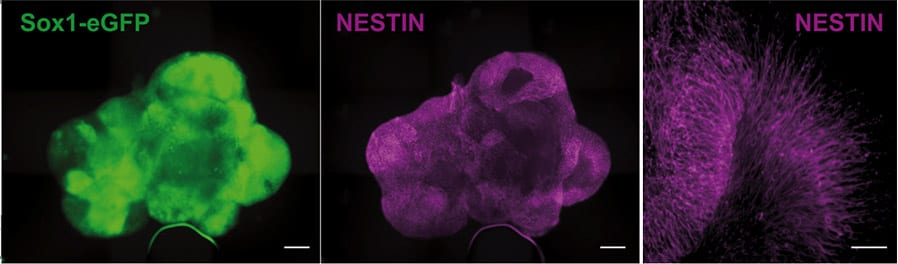
Day 2: Uniform spherical aggregates
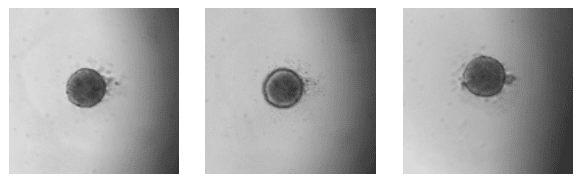
Day 4: Ruffling, indicating formation of vesicles
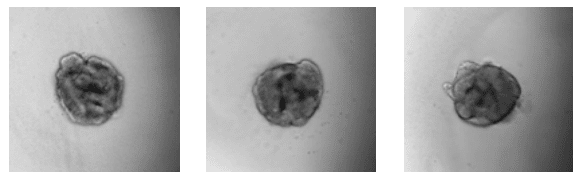
Fig. Aggregation and early organoid development of inner ear organoids on U-bottom Lipidure®-Coat Wells – courtesy of the Hashino lab, Indiana University School of Medicine