BL21 Gen-X™ Competent E.coli Expression Kit
More of your recombinant proteins with less background
The BL21 Gen-X™ E. coli is a next generation genetic variant of the standard BL21(DE3) strain. The BL21 Gen-X™ strain was created through careful cycles of mutagenesis, enrichment, and testing to provide a new and more powerful protein expression bacteria.
These special E. coli cells have a slower growth rate and characteristics that lead to significantly higher recombinant protein expression, while exhibiting much lower endogenous or background bacterial proteins. The results are clear, the BL21 Gen-X™ strain, along with the powerful Gen-X™ Induction Enhancer, will express more of what you want, and less of what you don’t in vivo.
Features
With the BL21 Gen-X™ Competent E. coli Expression Kit, there is now a better way to express your genes in bacteria: higher yields and lower amounts of contaminant protein will provide you with easier downstream purification and processing, saving you significant time and money.
- Up to 10X greater expression of your recombinant protein
- Reduced background protein levels
- Use to detect proteins not expressed or detected using other popular BL21 cells or kits
- Simple and straightforward protocol
- Compatible with all T7 promoter-based expression vectors
Applications
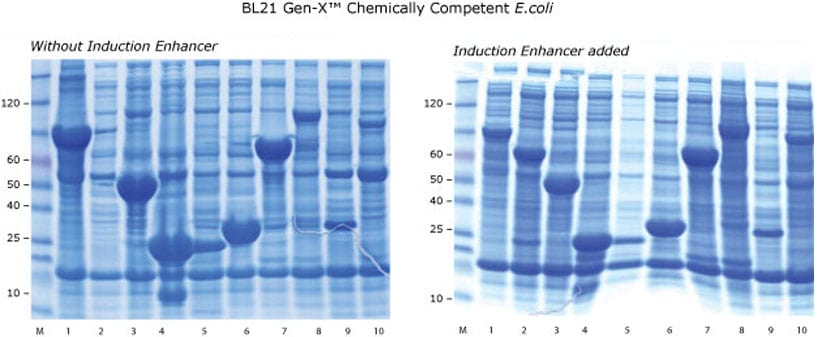
Cells were grown and induced as described in Figure 1. Additionally, 220ul of the 10X Gen-X™ Induction Enhancer were added at the time of IPTG addition; downstream processing was the same.
For many constructs, the addition of the Gen-X™ Induction Enhancer noticeably reduced background protein expression. This is because the Gen-X™ Induction Enhancer further reduces the growth rate of the cells. In some instances, there is a decrease in absolute recombinant protein expression.
However, the net result is a protein yield of higher purity due to the relatively lower background. In other cases, the Gen-X™ Induction Enhancer is essential for abundant target protein expression; for example in Lane 2, the 60KDa recombinant protein was observed only in the BL21 Gen-X™ cells along with the Gen-X™ Induction Enhancer, while in lane 8, the induction enhancer was responsible for increased expression of the 69 KDa protein.
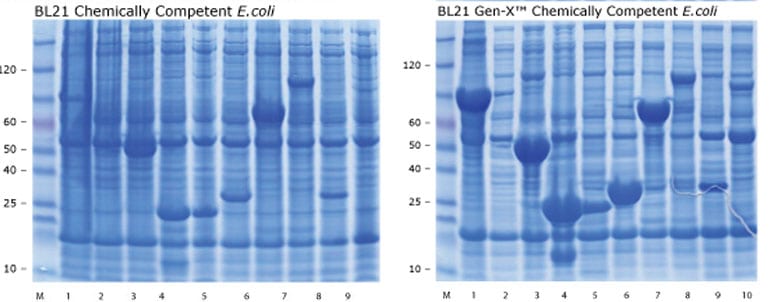
Ten 2 ml cultures of BL21 (DE3) each containing a different protein expression construct were grown at 37oC in LB and induced with 1 mM IPTG at OD600 = 0.4.
Cells were incubated overnight and 1 ml harvested. Concurrently, 2 ml cultures of BL21 Gen-X™ containing the same ten protein expression constructs were grown at 37oC in M9 minimal media and induced with 1 mM IPTG at the same OD.
Cells were incubated overnight and 1ml harvested. For both strains, pellets were suspended in 100ul, lysed, and 10ul electrophoresed on 4-12% polyacrylamide gels.
In lanes 1,3,4,5,6,8,9, and 10, the quantity of recombinant protein obtained from BL21 Gen-X™ cells was considerably greater and the background of endogenous E. coli proteins significantly lower