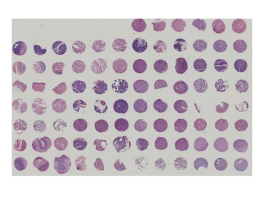
FDA Tissue Arrays
Want cost effective, time efficient, and quality-assured tissue arrays designed for FDA regulatory compliance for your TCR studies?
CollaFibR™ Type I Collagen Fibers
Dry Spun collagen fibers for 3D cell culture and bioprinting
Click to find out more

Need StemFit for GMP Manufacturing?
StemFit Basic04 CT is now available in a GMP-Compliant formulation!