Proteolysis Targeting Chimeras - PROTACs
A new and promising therapeutic method in drug discovery
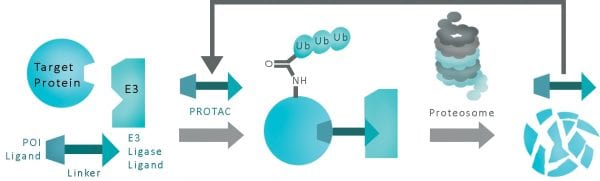
Targeted protein degradation using Proteolysis Targeting Chimeras, or PROTAC, is a new and promising therapeutic method in drug discovery. PROTACs have the ability to regulate protein degradation through targeted control of ubiquitin E3 ligases. This novel technology allows for the degradation of disease-related proteins and offers many advantages over traditional protein inhibition.
Our new range of critical enzymes, small molecular inhibitors, and homogeneous proximity assay services give you the power to research targeting protein degradation using PROTACs.
Ubiquitination
Ubiquitination is a key cellular function used to mark proteins for degradation by the proteasome. The protein of interest is labelled with one or many ubiquitin proteins. This process occurs in a sequential manner through three enzymes; ubiquitin activating enzymes (E1), ubiquitin conjugating enzymes (E2), and ubiquitin ligases (E3). The E3 ligases are necessary for the final transfer of ubiquitin to the protein of interest and occur either through a non-covalent or a covalent bond. There are over 6000 different E3 ligases that can mediate this process and are specific to the target protein being labelled for degradation.
PROTACs
PROTACs function by hijacking the final action of E3 ligases and subsequently tagging the protein of interest for ubiquitination. A PROTAC is composed of three parts; an E3 ligand that binds to an E3 ligase, a ligand that binds to the protein of interest, and a linker connecting the two ligands. When each ligand binds its respective partner, it triggers ubiquitination of the protein of interest. The PROTAC itself is not degraded by this process, making PROTACs a valuable tool for mediating protein degradation. A single PROTAC can promote the degradation of many subsequent proteins and functions substoichiometrically.
Advantages
The PROTAC method offers many advantages compared to traditional protein inhibition. Inhibitors require sustained protein binding to evoke the intended biological reaction. This can be problematic in the incidence of target overexpression, the presence of competing ligands, or protein mutations that lead to binding resistance. The PROTAC model bypasses these issues by promoting degradation that circumvents the native resistance of proteins against sustained inhibition.
Even select weak binding and promiscuous ligands can be utilized with PROTACs and still demonstrate high degradation efficacy. Also, ineffectual ligands which do not modulate the cellular functions of the protein of interest can be used to mediate degradation through PROTACs. Whether the ligand is a strong inhibitor, a weak binder, promiscuous, or ineffectual, there are many potential target proteins to utilize with PROTAC technology. In other words, PROTACs could have the ability to degrade proteins previously believed to be 'undruggable' through conventional small molecule inhibition.
| Name | Packsize | Order |
|---|---|---|
| (+)-JQ1 | 1 mg | View |
| (+)-JQ1 | 10 mg | View |
| (+)-JQ1 | 5 mg | View |
| (-)-JQ1 | 25 mg | View |
| (-)-JQ1 | 5 mg | View |
| ALK , GST-tag | 10 ug | View |
| BTK, GST-tag | 10 ug | View |
| CUL2, FLAG-Tag | 20 ug | View |
| HER2, GST-tag | 10 ug | View |
| NEDD4, FLAG-tag | 20 ug | View |
| PROTAC® Optimization Kit for BET Bromodomain-Cereblon Binding | 384 reactions | View |
| RIPK2, GST-tag | 10 ug | View |
| RediSolutionâ„¢ (+)-JQ1 | 1 mg | View |
| Tau-352 Protein | 20 ug | View |
| Tau-383 (N352H) Protein | 20 ug | View |
| Tau-383 Protein | 20 ug | View |
| Tau-412 Protein | 20 ug | View |
| Tau-441 (A152T) Protein | 20 ug | View |
| Tau-441 (K257T) Protein | 20 ug | View |
| Tau-441 (L266V) Protein | 20 ug | View |
| Tau-441 (N279K) Protein | 20 ug | View |
| Tau-441 (P301L) Protein | 20 ug | View |
| Tau-441 (P301S) Protein | 20 ug | View |
| Tau-441 (R406W) Protein | 20 ug | View |
| Tau-441 (S198A) Protein | 20 ug | View |
| Tau-441 (S214A) Protein | 20 ug | View |
| Tau-441 (S305N) Protein | 20 ug | View |
| Tau-441 (dK280) Protein | 20 ug | View |
| Tau-441 (dN296) Protein | 20 ug | View |
| c-CBL TR-FRET Assay Kit | 384 reactions | View |
Homogeneous Proximity Assays
We offer a homogeneous proximity assay service to assess the chemical adaptor function of PROTACs. If you are interested in this service, contact us for more information.