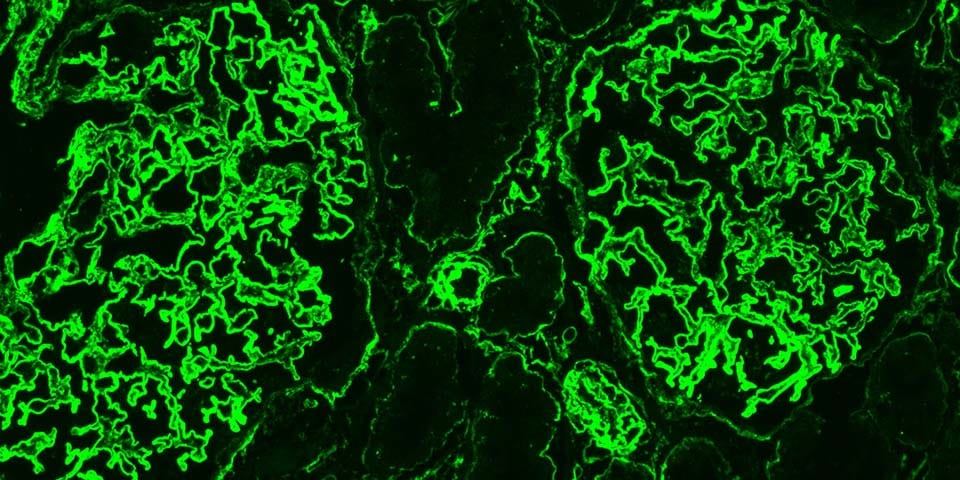
Heparan Sulfate (HS) is a linear polysaccharide that is attached to a core protein to form HS Proteoglycans on the cell membrane or in the extracellular matrix. HS is emerging as a key factor in infection by the SARS-CoV-2 virus, which is the cause of the current COVID-19 Coronavirus pandemic. Multiple mechanisms underlying the pathogenesis of COVID-19 involve HS, such as cellular infection, inflammation and vascular leakage due to heparanase-mediated degradation of HS in the endothelial glycocalyx. AMSBIO’s offering of Heparan Sulfate antibodies, along with enzymes, standards and kits therefore provides an important resource for understanding the pathogenesis of COVID-19, and so combatting the pandemic.
A Novel Cell Therapy for Liver Cirrhosis with Human Bone Marrow Derived MSCs
December 14, 2020
How to Get Your Organoids Through Lockdown
October 21, 2020In particular, the JM403 Heparan Sulfate antibody from AMSBIO has been used in two novel assays developed and applied by Prof. Dr. Johan van der Vlag and colleagues at the Radboud University Medical Center, Nijmegen, Netherlands. They developed a JM403-based plasma heparanase activity assay, and a JM403-based HS competition ELISA to measure plasma HS levels. Notably, AMSBIO is also the source of RaZie ready-to-use kits to measure Heparanase activity in cell culture supernatants, biological fluids and tissue samples.
The JM403 antibody appeared an unique and powerful tool to develop the new assays to measure plasma heparanase and HS levels in patients with COVID-19. A particular advantage of JM403 is that it is not reacting with heparins, which would have been a disturbing factor in our analyses, and which now allowed us to evaluate the effect of treating COVID-19 patients with low molecular weight heparins (LMWH).
Prof. Dr. Johan van der Vlag, Radboud University Medical Center, Nijmegen, Netherlands

Heparan Sulfate in SARS-CoV-2 Infection
Heparan Sulfate has been established as a necessary co-factor for infection of cells by SARS-CoV-2: with the viral spike protein interacting with both cellular Heparan Sulfate and Angiotensin-Converting Enzyme 2 (ACE2) through its receptor-binding domain (RBD) – see Clausen, Eso et al (2020) below.
Heparan Sulfate and Heparanase Enzyme in COVID-19 Disease
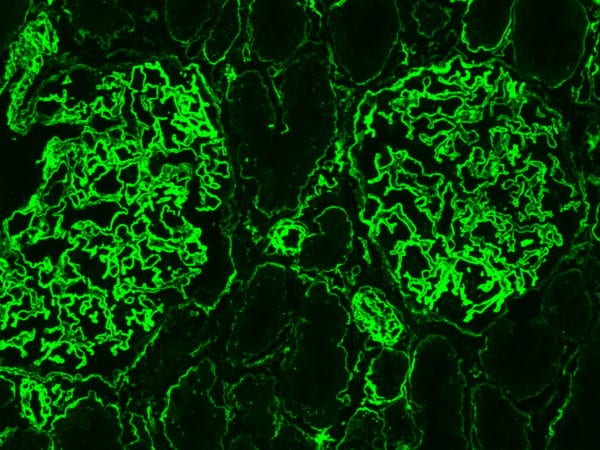
Heparan Sulfate also forms a key component of the glycocalyx – a complex mixture of glycans and glycoconjugates surrounding all cells. The integrity of the endothelial glycocalyx is crucial for endothelial barrier function, especially in the lungs and kidneys – and disruption of this barrier is a feature of many of the more serious symptoms of COVID-19 disease – notably acute respiratory distress syndrome (ARDS), acute kidney injury and proteinuria (leakage of protein into the urine).
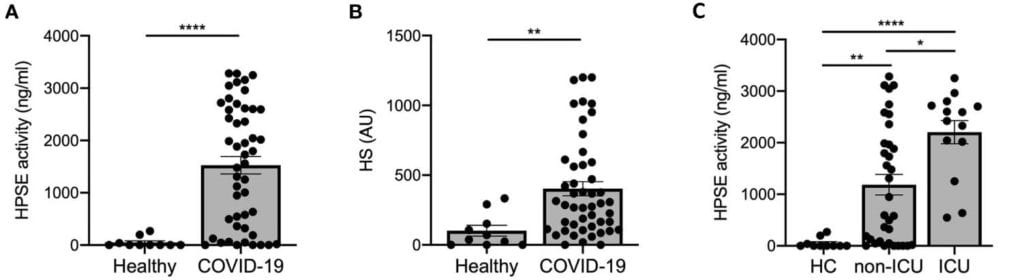
This disruption of the endothelial barrier may be mediated by Heparanase (HPSE), the only known mammalian enzyme capable of degrading HS, as both HPSE activity and HS levels are significantly elevated in the plasma of COVID-19 patients compared to healthy controls (Fig 1 A, B). Further, plasma Heparanase activity is also associated with COVID-19 disease severity, with intensive care unit (ICU) patients on mechanical ventilation showing significantly higher levels than non-ICU patients, who in turn had higher activity than healthy controls (Fig 1C).
| Product Code | Product Name | Pack Size | Datasheet | Order Now |
|---|---|---|---|---|
| 370730-S | Ab-Heparan Sulfate (JM-403 epitope), Purified | 50 ug | View | Order Now |
| 370730-1 | Ab-Heparan Sulfate (JM-403 epitope) / Rat, (JM-403) (mo) (M), Purified | 0.2 mg | View | Order Now |
| 370255-S | Ab Heparan Sulfate, purified (clone F58-10E4) | 50 ug | View | Order Now |
| 370255-1 | Ab Heparan Sulfate, purified (clone F58-10E4) | 0.2 mg | View | Order Now |
| Ra001-BE-K | Heparanase Assay Kit with Enzyme as Positive Control | 96 reactions | View | Order Now |
| Ra001-02-K | Heparanase Assay Kit without Positive Control Enzyme | 96 reactions | View | Order Now |
References
Increased plasma heparanase activity in COVID-19 patients
Buijsers, B., Yanginlar, C., de Nooijer, A., Grondman, I., Maciej-Hulme, M. L., Jonkman, I., … & van der Vlag, J. (2020) Frontiers in Immunology, 11, 2572. Citing JM403 antibody.
Beneficial non-anticoagulant mechanisms underlying heparin treatment of COVID-19 patients
Buijsers, B., Yanginlar, C., Maciej-Hulme, M. L., de Mast, Q., & van der Vlag, J. (2020) EBioMedicine, 59, 102969.
SARS-CoV-2 Infection depends on cellular Heparan Sulfate and ACE2
Clausen, T. M., Sandoval, D. R., Spliid, C. B., Pihl, J., Perrett, H. R., Painter, C. D., … & Esko, J.D. (2020) Cell (in press). Citing 10E4 antibody.
Additional Resources
We couldn't find any records.
Related Products
Subscribe to our newsletter
Be the first to hear about upcoming events, product news, special offers, application notes, and more