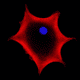
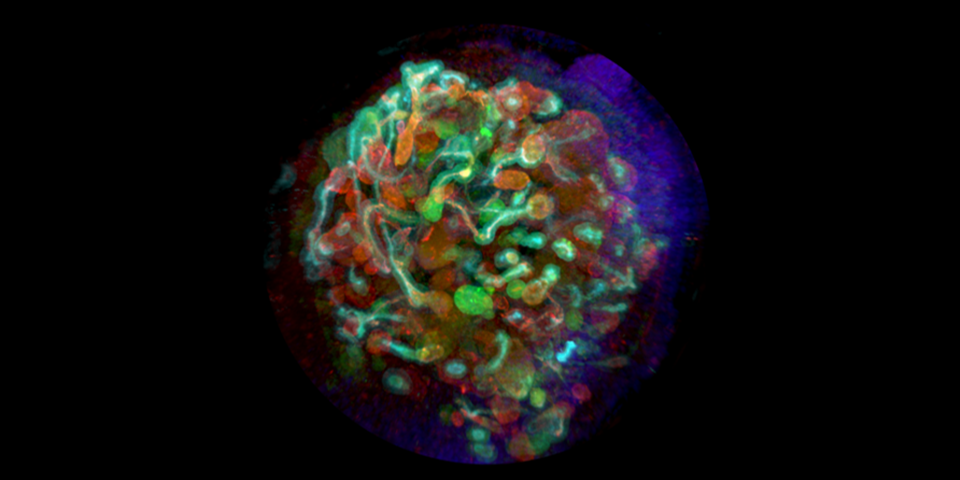
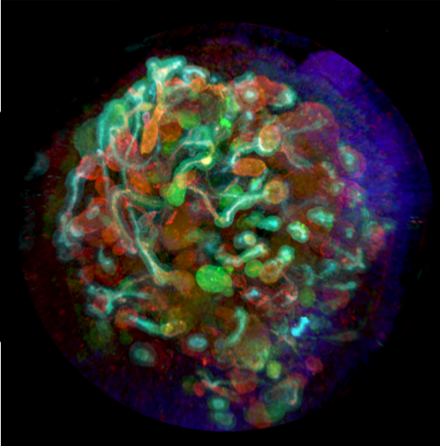
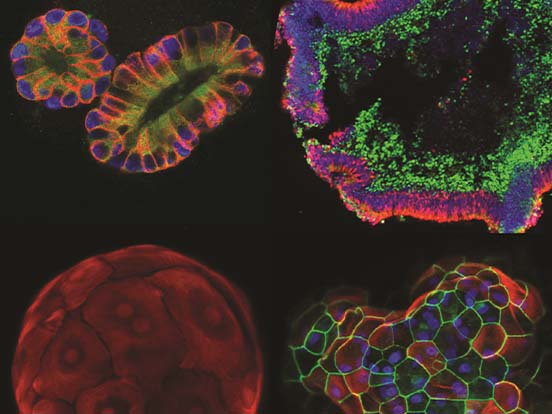
Professor Ryuji Morizane, a renowned expert in renal research at Harvard Medical School, discusses his award-winning research* where organoid and bioengineering technology was successfully combined to create a kidney organoid with vascular structure.
To help facilitate this pioneering work, Professor Morizane’s research group used StemFit® – a feeder-free culture medium for embryonic (ES) and induced pluripotent stem cells (iPSC) without human or animal-derived components.
*Professor Morizane’s research was selected for the NIH Director’s New Innovator Award in 2019.