Hugues Lortat-Jacob and his team of scientists at Institut Biologie de Structurale (IBS) in Grenoble stand out as key opinion leaders in the research community of glycobiology. Although abundant and integral to many cellular processes, the full breadth of the role that glycosaminoglycans (GAGs) and proteoglycans play in physiology and pathology is not yet understood. The research group led by Lortat-Jacob at IBS has made significant strides in advancing the field, exploring the structure and function of GAGs and proteoglycans present at the cell-extracellular matrix interface. Lortat-Jacob and his colleagues seek to reveal the cellular processes involved in determining their structure while simultaneously decoding how GAGs – such as heparan sulfate – regulate protein activities. By unravelling the intricacies of GAG activity, Lortat-Jacob and his colleagues have illuminated the contributions of GAGs to a diverse range of biological mechanisms at an atomic level. Their findings bring us closer to understanding biological phenomena such as cancer, cellular defences against infection, DNA repair, and more.

Investigating Glycans – shedding new light on the ‘dark matter’ of biology
January 10, 2023
iPS Cell Based Therapies for Genetic Skin Disease
August 31, 2022Studies by the researchers at IBS have provided valuable insight into the implications of heparan sulfate in several diseases. Recently, heparan sulfate and syndecan-1 were identified as potential clinical biomarkers of COVID-19 and multisystem inflammatory syndrome in children (MIS-C) – a rare complication of COVID-19 resulting in immune dysregulation and multiple organ dysfunction. Researchers in Lortat-Jacob’s group have also explored the molecular mechanisms behind the biosynthesis of heparan sulfate, a process catalysed by enzymes EXT1 and EXT2 which was previously not understood. By revealing how EXT1 and EXT2 synthesise heparan sulfate, the team at IBS have laid the groundwork for future research on the enzymes’ potential roles in disease pathophysiology.
At AMSBIO we’re proud of our longstanding association with Hugues Lortat-Jacob and his colleagues at IBS, supplying their scientists with heparan sulfate and heparan sulfate -detecting antibodies for their research. The Lortat-Jacob Team also uses cryopreservation medium CELLBANKER 2, provided by AMSBIO, to ensure reliable long-term storage of their cell samples.
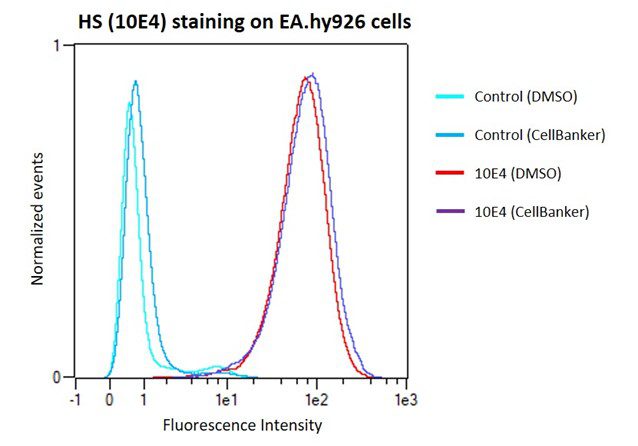
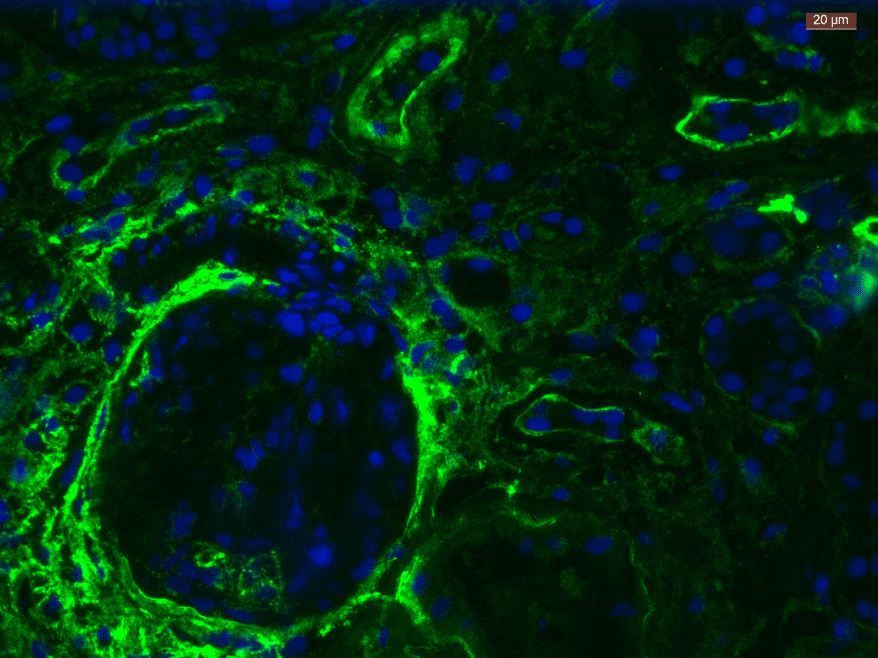
Figure 1. Heparan Sulfate (HS/10E4) level measured on EA.hy926 cells via fluorescence intensity, demonstrating intact expression using CELLBANKER 2 compared to DMSO. Results courtesy of Rabia Sadir, Lortat-Jacob Team (Structure and Activity of Glycosaminoglycans Group), IBS, France
“The advantage of using CELLBANKER is the time-saving.” – Rabia Sadir, Lortat-Jacob Team (Structure and Activity of Glycosaminoglycans Group), IBS, France
As illustrated in the figure above, Rabia Sadir and colleagues of the Lortat-Jacob team at IBS found that EA.hy926 cells that were cryopreserved using CELLBANKER 2 maintained their heparan sulfate expression post-thaw. Unlike DMSO and other cryopreservation mediums, CELLBANKER 2 can be frozen directly at 80°C – saving Sadir and the team time and reducing labour, while preserving cells to a consistently high standard for up to 10 years. Sadir and her colleagues used 10E4 antibody to quantify the heparan sulfate expression on the EA.hy926 cell surfaces – one of three heparan sulfate antibodies available from AMSBIO (10E4, 3G10, and JM403).
References
- Structure of the human heparan sulfate polymerase complex ext1-ext2. Leisico, F., Omeiri, J., Le Narvor, C., Beaudouin, J., Hons, M., Fenel, D., Schoehn, G., Couté, Y., Bonnaffé, D., Sadir, R., Lortat-Jacob, H., & Wild, R. (2022). Nature Communications, 13(1). https://doi.org/10.1038/s41467-022-34882-6
- Heparanase is a putative mediator of endothelial glycocalyx damage in COVID-19 – a proof-of-concept study. Drost, C. C., Rovas, A., Osiaevi, I., Rauen, M., van der Vlag, J., Buijsers, B., Salmenov, R., Lukasz, A., Pavenstädt, H., Linke, W. A., & Kümpers, P. (2022). Frontiers in Immunology, 13. https://doi.org/10.3389/fimmu.2022.916512
- Heparan sulfate differentially controls CXCL12α- and CXCL12Γ-mediated cell migration through differential presentation to their receptor CXCR4. Connell, B. J., Sadir, R., Baleux, F., Laguri, C., Kleman, J.-P., Luo, L., Arenzana-Seisdedos, F., & Lortat-Jacob, H. (2016). Science Signaling, 9(452). https://doi.org/10.1126/scisignal.aaf1839