BioPORTER® Protein Delivery Reagent
Direct translocation of proteins into living cells
BioPORTER is a lipid-based protein delivery system that efficiently transports proteins into living cells. It is a unique cationic lipid mixture that interacts non-covalently with the protein, creating a protective vehicle for immediate protein delivery. BioPORTER makes studying protein functionality faster and easier than ever before by bypassing the traditional DNA transfection, transcription, and protein translation processes.
Features
It is very effective in delivering a number of proteins, peptides, and other macromolecules into the cytoplasm of many different cells, retaining the structure of the proteins whilst leaving the transduced cells unharmed. The BioPORTER® Reagent is especially useful when studying protein function in cells that are difficult to transfect using traditional DNA transfection reagents, such as human dendritic cells.
- Effective in a variety of different cell types
- Can be used to deliver a range of different macromolecules into cells
- Can be used to deliver fluorescently labeled antibodies into cells
- Fast and easy protocol
How does it work?
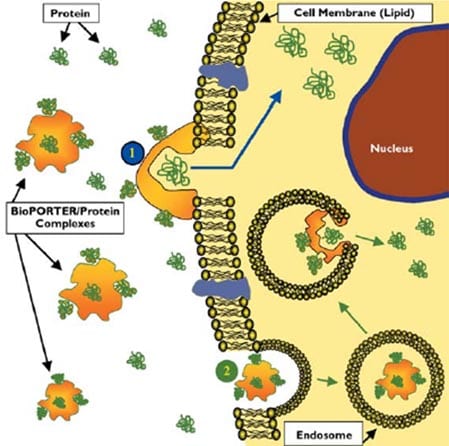
Once the positively charged BioPORTER/protein complexes are applied to cells, they attach to the negatively charged cell surface. The BioPORTER reagent fuses directly with the plasma membrane and the captured protein is internalized into the cell, or alternatively, the BioPORTER/protein complexes are endocytosed and fuse with the endosome, releasing the captured protein into the cytoplasm (Figure 1).
Due to the simple delivery mechanism and because there is no fusion partner or covalent bond formation, BioPORTER can efficiently deliver functionally active proteins into a wide range of cell types.
Easy-to-use System
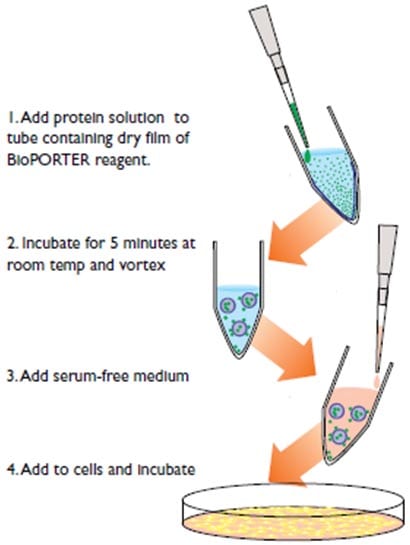
Figure 2 shows the protocol for using BioPORTER. Simply add the protein solution to a tube containing a dry film of BioPORTER reagent, incubate for 5 minutes, add the serum free medium, and you are ready to apply the mixture to cells for protein delivery. It’s that easy.
Single Use or High Throughput: For maximum speed and convenience, use the new BioPORTER QuikEase™ Kit. Each QuikEase kit contains 96 single reaction size vials that are pre-coated with BioPORTER reagent. You can either separate each vial for single use or use the entire pack for high-throughput experiments. The BioPORTER QuikEase kit saves hours of time by eliminating the need to coat the BioPORTER® Reagent onto individual vials.
Applications using BioPORTER Protein Delivery Reagent
- Apoptosis
- Cell cycle control
- Protein localization
- Transcriptional regulation
- Intracellular signal pathways
- Peptide library screening
- Expression monitoring
- Protein complementation
Protein Functionality
When delivering proteins with BioPORTER protein delivery reagent there’s no fusion partner or covalent bond formation, allowing the translocated protein to remain functionally intact.
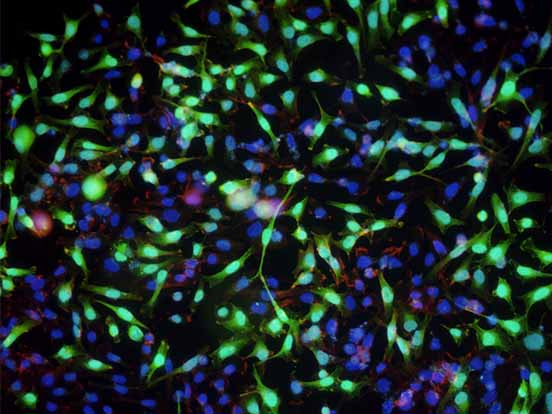
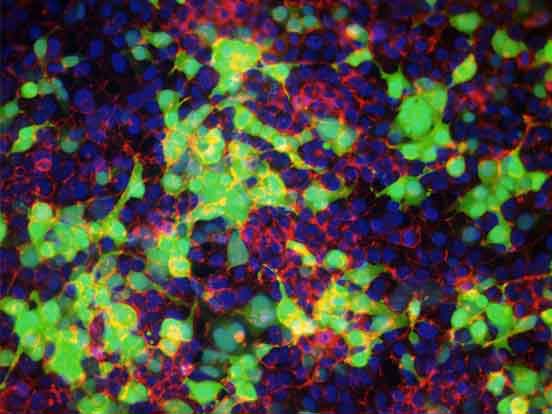
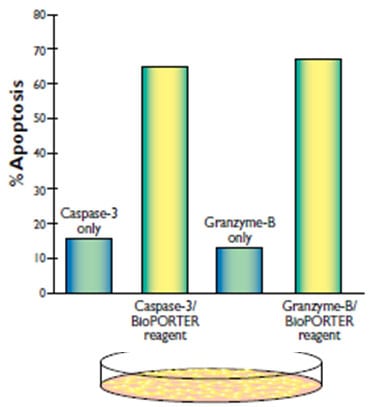
To demonstrate, active recombinant human caspase-3 and granzyme-B were delivered into Ki-Ras 267 β1 prostate cancer cells using BioPORTER. Figure 3 shows that both proteins are functionally active after delivery and can successfully induce apoptosis.
For studies in which protein functionality is key, such as apoptosis, cell cycle control, and protein complementation, BioPORTER has proven to be an easy and effective tool
Proteins and Cell Lines Successfully Tested with BioPORTER Reagent
| Proteins Delivered | Cell Lines Tested |
|---|---|
| Fluorescently Labelled Antibodies High MW Dextran Low MW Dextran Phycoerythrin-BSA (MW-300kD) Caspase 3 Caspase 8 β-galactosidase |
HeLa-S3 COS-7 BHK-21 B16-F0 Actuve Myeloctic Leukemia CHO-K1 293 MDCK COS-1 RAW Macrophage NIH-3T3 Jurkat P19 CV-1 Ki-Ras 267 β1 Chronic Myelocytic Leukemia |
Storage
The BioPORTER® Protein Delivery Reagent is shipped frozen. For maximum stability, store all reagents at -20°C upon receipt. BioPORTER® reagent is stable for 1 year when stored at the recommended temperature.
Citations
Cellular loading of NGF, a potential therapeutic protein, in primary macrophages
Bottger, D.,et al (2010) Brain Res 2 1312: 108.
Apoptosis pathway elucidation in primary mouse cortical neurons
Alano, C.C.,et al (2010) J. Neurosci. 30(8): 2967-2978.
Parkinson's Disease pathway elucidation in QBI-HEK-293 cells
Luk, K.C., et al (2009) PNAS. 106(47): 20051-20056.